VIVOTIF may help provide protection against typhoid fever for at least 5 years.1


- Not all recipients of VIVOTIF will be fully protected against typhoid fever1
- Vaccinated individuals should continue to take personal precautions to avoid contact or ingestion of potentially contaminated food or water1
- The optimum booster schedule for VIVOTIF has not been determined1
- It is recommended that a re-immunization dose consisting of 4 vaccine capsules taken on alternate days be given every 5 years under conditions of repeated or continued exposure to typhoid fever1
Sampling local cuisine can be one of the most enjoyable parts of traveling; however, when it comes to food and drink, you should understand the risk that typhoid fever can present.
Typhoid fever is a potentially severe and life-threatening illness caused by a type of bacteria called Salmonella typhi.2 It’s spread by contaminated food and water.2 Common symptoms of typhoid fever include loss of appetite, headache, weakness, stomach pain, and constipation or diarrhea.3,4 In severe cases, stomach bleeding, brain damage, and death may occur.4
Typhoid fever is commonly found in regions of the world where water and food may be unsafe, including parts of South Asia, Africa, Latin America, Southeast Asia, East Asia, and the Caribbean.4,5

Take precautions even after vaccination because the vaccine does not completely protect against typhoid fever. While traveling, follow good hygiene practices and ensure the food and water you consume is as safe as possible.1
It’s also important to know that the danger from typhoid fever doesn’t end when symptoms disappear.3 Even when symptoms seem to go away, you may still be carrying Salmonella Typhi and can pass it on to someone else.3
VIVOTIF is the only FDA-approved oral vaccine for typhoid fever.1,4
VIVOTIF is a prescription vaccine you take by mouth to help protect you from typhoid fever.1 VIVOTIF is for adults and children greater than 6 years of age.1
Routine typhoid vaccination is not recommended in the United States of America. Selective immunization against typhoid fever is recommended for the following groups: 1) travelers to areas where there is a recognized risk of exposure to S. typhi, 2) persons with intimate exposure (e.g. household contact) to an S. typhi carrier, and 3) microbiology laboratorians who work frequently with S. typhi. There is no evidence to support the use of typhoid vaccine to control common source outbreaks, disease following natural disasters, or in persons attending rural summer camps.
Not all recipients of VIVOTIF will be fully protected against typhoid fever. Vaccinated individuals should continue to take personal precautions against exposure to typhoid organisms. The vaccine will not afford protection against species of Salmonella other than Salmonella typhi or other bacteria that cause enteric disease. The vaccine is not suitable for treatment of acute infections with S. typhi.

While typhoid fever can be treated with antibiotics, according to data reported in 2017, 74% of Salmonella Typhi infections are resistant to one of the most commonly used antibiotics.6
VIVOTIF can help provide protection
when the full course is taken at least 1 week prior to traveling.1
VIVOTIF is an oral vaccine.1
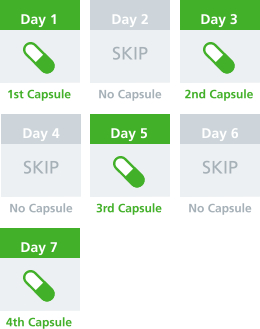
How to take VIVOTIF1:
- One complete dosing schedule consists of 4 capsules
- VIVOTIF is taken every other day for 7 days
(see illustration below)
GET TEXT REMINDERS
To enroll in the TAKE 4 text reminder program, text "TRAVEL" to 1-833-207-3377 on the day of your first dose. You will receive a text every other day reminding you to take your next dose and to refrigerate the remaining capsules.*
*The information requested will be used by Bavarian Nordic to send you information related to vaccines and travel health. Message and data rates may apply. This is a recurring program that includes a number of messages over the course of 7 days. At any time during the program, patients may text "HELP" for assistance or "STOP" to discontinue receiving messages.
All 4 doses of VIVOTIF should be taken at least 1 week before traveling.1

Swallow one capsule about 1 hour before a meal with a cold or lukewarm (temperature not to exceed body temperature, e.g., 37°C [98.6°F]) drink.1 It doesn’t matter what time you take your capsule, as long it’s at least 1 hour before a meal1:
- Swallow the capsule as soon after placing in the mouth as possible. Don't open or chew capsule
- Do not freeze the capsules. Keep capsules refrigerated (35.6ºF–46.4ºF) until you're ready to take them
GET TEXT REMINDERS
To enroll in the TAKE 4 text reminder program, text "TRAVEL" to 1-833-207-3377 on the day of your first dose. You will receive a text every other day reminding you to take your next dose and to refrigerate the remaining capsules.*
*The information requested will be used by Bavarian Nordic to send you information related to vaccines and travel health. Message and data rates may apply. This is a recurring program that includes a number of messages over the course of 7 days. At any time during the program, patients may text "HELP" for assistance or "STOP" to discontinue receiving messages.
ASK HOW VIVOTIF MAY HELP PROTECT YOU WHILE TRAVELING
TO TYPHOID FEVER–AFFECTED AREAS.



